
In IVF laboratories, traceability is directly linked to patient safety. A single undocumented consumable lot or mismatch in records can compromise investigation, delay corrective action, and ultimately affect clinical outcomes.
Integrating Allwin consumables with MIRI® Evidence, an RFID-based witnessing and traceability system designed for IVF laboratories, provides an end-to-end solution for consumable tracking, documentation, and performance monitoring.
Why Lot Management Matters in IVF
Each IVF cycle involves multiple consumables, including ovum pickup needles, embryo transfer catheters, IUI catheters, denudation pipettes, and vitrification devices. Every product carries a lot number that links it to a specific manufacturing batch. Proper lot management ensures full traceability from manufacturer to patient, rapid recall response in case of quality alerts, compliance with regulatory requirements, and clear documentation for audits and inspections.
Manual recording of lot numbers in paper logs or spreadsheets increases the risk of transcription errors and incomplete records. As laboratories manage higher cycle volumes and stricter regulatory expectations, digital lot tracking becomes increasingly important.
Allwin Consumables: Quality and Consistency for ART
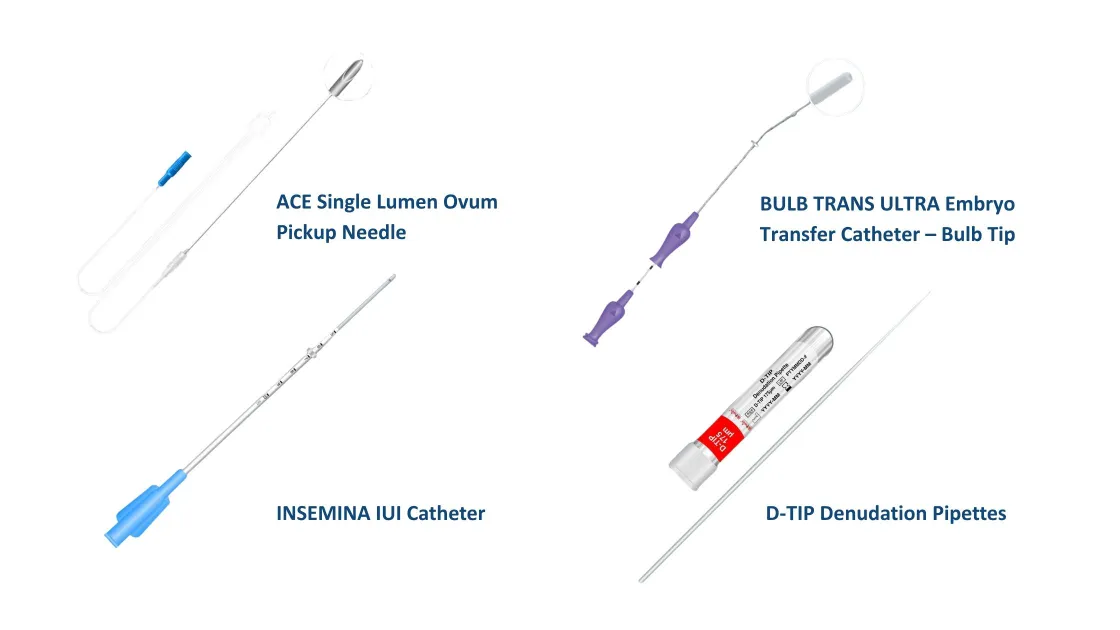
Allwin consumables are developed specifically for assisted reproductive technology procedures. The portfolio supports the full IVF workflow and includes:
- Ovum pickup needles in single and double lumen configurations, with echogenic tips to improve ultrasound visibility and support precise follicle aspiration
- Embryo transfer catheters including Bulb Trans Ultra featuring super thin inner catheters with optional metal guide. Bulb-shaped tip helps reduce trauma and bleeding, while Pro Echo Trans and Pro Pass Echo Trans, full-length echogenic tip supports precise ultrasound-guided placement
- IUI catheters designed for controlled insertion, featuring straight, curved tip, and trackable malleable stylet options, tested with Human Sperm Survival Assay (HSSA) and Limulus Amebocyte Lysate (LAL) for biocompatibility
- Denudation pipettes for precise oocyte handling and ICSI preparation, offered in a wide range of sizes (135–300 µm) to suit different laboratory needs.
- Vitrification devices validated through MEA (Mouse Embryo Assay) and LAL (Limulus Amebocyte Lysate) testing, and gamma sterilized to ensure biological safety and sterility
All products are sterile, single-use, quality-controlled devices developed specifically for ART laboratory standards.
Each product is clearly labeled with lot number and expiry date. When incorporated into a digital workflow, this information becomes part of a complete electronic traceability record.
MIRI® Evidence: RFID-Based Witnessing and Traceability

MIRI® Evidence is an RFID-based witnessing system designed to reduce manual errors and improve traceability throughout the IVF workflow.
At each critical step, consumables and patient identifiers are scanned using RFID readers and barcode scanner. The system records:
- Patient identity
- Procedure step
- Date and time
- Operator
- Consumable lot number
The result is a real-time, digital record that replaces manual documentation. The system can be integrated into laboratory workflows alongside existing equipment, supporting consistent verification without interrupting routine procedures.
Seamless Lot Management Workflow

Combining Allwin consumables with MIRI® Evidence allows IVF laboratories to implement a structured and data-driven lot management process that supports both compliance and clinical oversight.
- Receiving and stock entry — Consumables are logged with lot numbers and expiry dates before use.
- Procedure-level verification — Items are scanned at the point of use and linked to the correct patient.
- Automatic documentation — The system records lot numbers together with patient, operator, and procedure data.
- Recall and audit readiness — Laboratories can immediately identify all cases associated with a specific lot.
- Performance tracking and analysis — Consumable lots can be reviewed against clinical outcomes, allowing laboratories to identify trends and investigate performance variations.
From Traceability to Clinical Performance Monitoring
When consumable usage is linked to clinical outcomes, lot management becomes a clinical performance monitoring and decision-support tool.
Laboratories can compare fertilization rates, blastocyst development, or pregnancy outcomes across different consumable lots or suppliers. Performance before and after changing a brand can be evaluated using structured data.
This enables:
- Early detection of performance deviations
- Evidence-based evaluation of consumables
- More informed purchasing decisions
- Stronger internal quality discussions
Instead of relying on subjective feedback, laboratories can use real data to guide decisions.
Supporting Compliance and Laboratory Standards
Traceability of consumables is required under ISO 15189 and reinforced by ESHRE guidelines for good practice in IVF laboratories. ESHRE recommends that laboratories maintain a stock management system that records batch number, date of entry, and expiry date for all consumables and media.
The guidelines also emphasize the use of sterile single-use consumables, proper identification to avoid misuse, and ensuring that all products are used within their validated shelf life. These requirements support consistent quality control and enable laboratories to investigate any deviation in performance.
A Complete Solution for the Modern IVF Laboratory
Allwin consumables provide reliable, women's health-focused tools designed for clinical use. MIRI® Evidence adds a digital layer that ensures every consumable is verified, recorded, and linked to outcomes.
Together, they support a more structured, transparent, and data-driven IVF laboratory environment.
For laboratories looking to strengthen traceability and quality control, this integration offers not only compliance, but a clearer understanding of how consumables impact clinical results.
Learn more about Allwin consumables at www.allwinmedical.com
Explore MIRI® Evidence at www.esco-medical.com







