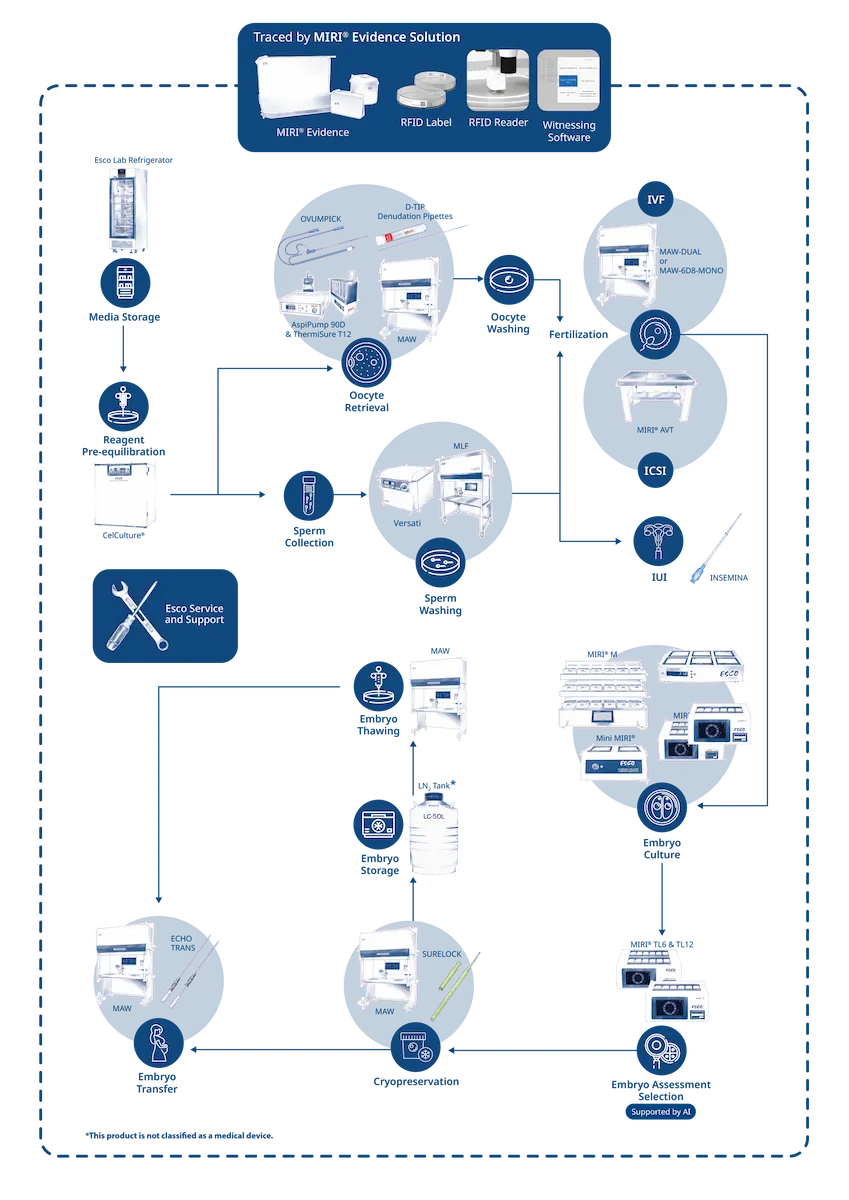
Workflow Categories
-
RFID Witnessing and Traceability System
-
Media Storage
-
Reagent Pre-equilibration
-
Oocyte Retrieval
-
Sperm Washing
-
IUI
-
Fertilization
-
Embryo Culture
-
Embryo Assessment & Selection
-
Cryopreservation
-
Embryo Storage
-
Embryo Thawing
-
Embryo Transfer
Products
Featured Products
-

MIRI® M Multiroom IVF Incubator
The Esco Medical MIRI® M Multiroom IVF Incubator is a modular system with up to 18 independent and movable chambers. Each chamber is designed to maintain a stable and secure environment even during movement, ensuring optimal protection for samples at all times. Its recirculating gas system, with a VOC/HEPA filter and UVC light, maintains optimal conditions, while precise temperature control enhances sample safety. The display provides visual patient confirmation, and built-in software ensures digital verification and tracking, making it a reliable choice for modern IVF laboratories.
-

MIRI® Evidence RFID Witnessing and Traceability System
MIRI® Evidence is redefining the way traceability in laboratory procedures is managed and stored in fertility clinics, eliminating errors, preventing system mix-ups, saving time, and helping clinics to comply with regulations. This IVF traceability tool provides a single, secure platform that ensures all patients are scheduled on time and documented properly - saving time and improving outcomes across the board.
-

MIRI® Time-Lapse Incubator
The MIRI® Time-Lapse Incubator is a multiroom incubator with a built-in camera and microscope. Designed and manufactured in EU, the MIRI® Time-Lapse Incubator provides high quality time-lapse images of embryos developing in "real-time" without having to remove the embryos from the safety of the incubation chamber for manual microscopy. Time-lapse embryo monitoring provides detailed morphokinetic data throughout embryo development, which is not available on routine spot microscopic evaluation. This allows all important events to be observed, helping to identify healthy embryos with the highest probability of implantation, with the aim of achieving higher pregnancy rates.
-

Animal IVF
Esco Animal IVF products are designed to meet the demands of IVF laboratory conditions...